Background
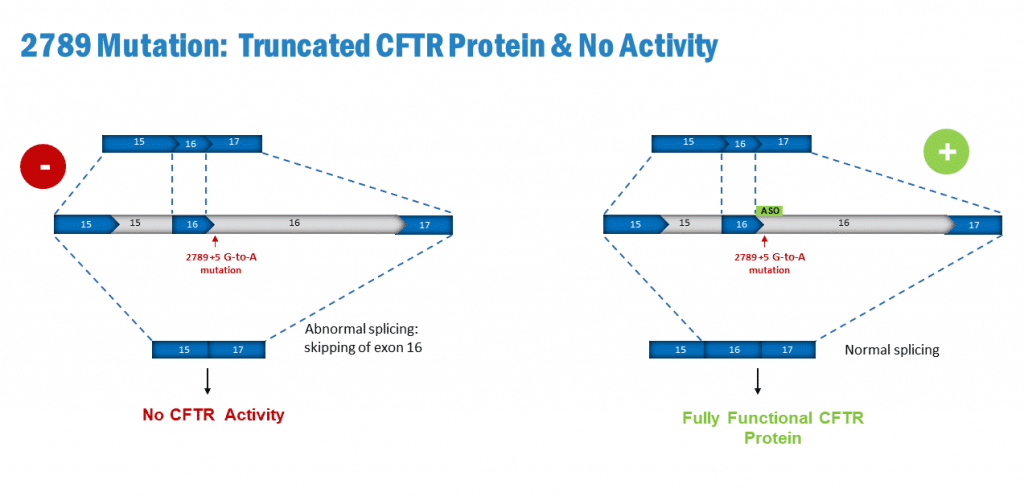
2789+5G->A is among identified mutations in the CFTR genes that generate both aberrant and correct splicing of the CFTR gene. It is classified as a class V mutation (mutations that lead to reduced amounts of normal CFTR proteins at the cell surface). The 2789 splicing mutation causes skipping over exon 16 leading to a frameshift and a premature termination codon.
This mutation is associated with sever lung disfunction usually above the age of 20.
Prevalence
There are ~1250 patients carrying at least one 2789 allele, listed in the US Cystic Fibrosis Foundation (CFF) and European Cystic Fibrosis Society (ECFS) patient registries.
Challenge
Although CFTR modulators are approved for patients carrying the 2789 mutation in the US, their effect is very minimal with no approved drug in the EU. Therefore, a treatment with higher efficacy is needed for patients carrying the 2789 mutation.
SpliSense Solution
SPL16 – ASO given by inhalation, penetrates the cells and avoids skipping over exon 16 in the CFTR transcript. This will avoid the maturation defect and proteasomal degradation of misfolded proteins generated by the 2789 mutation. Treatment with SPL16 leads to the generation of fully functioning WT CFTR .
The program is in pre-clinical stages.
